Chemical reactivity
Many smell molecules exhibit chemical reactivity.
This is partly the answer to why their smell is often so intense – because they stimulate the receptors of the olfactory epithelium, resulting in a strong psycho-physiological response. Aldehydes are also chemically reactive and it can be proven that some molecules responsible for, for example, “popular” bad odours will react with them to form new molecules with greatly diminished odour or odourless. The chemical reactivity of the aldehyde group -CHO has given rise to many processes in the modern chemical industry, mainly in the production of resins and plastics, as well as hygiene products and disinfectants.
The simplest aldehyde – formaldehyde HCHO is an important component of melamine, carbamide, and acetal resins. It is also a component of the first fully synthetic plastic – bakelite, a phenol-formaldehyde resin. In addition, formaldehyde is still a widely used disinfectant and fumigant (a disinfectant that emits smoke).

For our purposes, however, formaldehyde is useless. It is over-reactive, has a strong, pungent, and irritating odour (acts on the trigeminal nerve endings), and is otherwise poisonous. However, many other aldehydes are known and used. Almost all of them have an interesting smell and are widely used in the production of fragrance and flavour products. We have found that some aldehydes enter into stable chemical reactions with a broad group of odour-active compounds, as well as other smell compounds that do not evoke unambiguously negative associations (e.g., coffee aroma). A result is a new tool in the fight against noxious odours, and the areas of application are limitless.
Our neutralizers are a mixture of at least two aldehyde neutralizers, wherein one has an unsaturated bond on the carbon at the alpha position of the CHO group and the other does not. The active ingredients in the preparations act through nucleophilic addition or substitution reactions with nitrogen and sulphur on the carbon of selected aldehydes and ketones. The resulting reactions are irreversible. This idea was developed as it was found that certain pairs of aldehydes together work more effectively in binding odours than if they were to react separately. Following this discovery, we found more such relationships with min. 2 and more combinations of aldehydes, ketones and ketoalkynes.

In many cases, the reaction mechanism has not been completely understood and described. Empirically, we are able to demonstrate that such vapours function and increase the effectiveness of the neutralizing mixture in reducing the concentration of undesirable substances in the air.
In the area of the newest generation of neutralizers, we also deal with the addition (attachment) reaction with breaking the multiple carbon-carbon bonds. In general, these reactions are neither fast nor complete in 100% until the neutralizing components appear in sufficient excess to push the reactions toward completion.
The following are examples of selected reactions of aldehydes with odours containing thiol and amine groups.
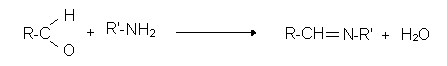
Example of the reaction of an aldehyde with a simple primary amine:
RCHO + R1NH2 → RCH = NR1 + H2O
Example of the reaction of an aldehyde with mercaptan:
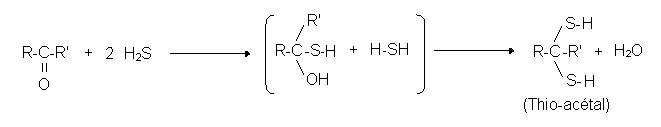
R1CHO + 2R2SH → R1CH(SR)2 + H2O
In both cases, the resulting molecules are generally less volatile, and the osmophoric nitrogen (N) or sulphur (S) atoms are effectively “hidden” in the molecule and no longer have as much power to irritate olfactory receptors.
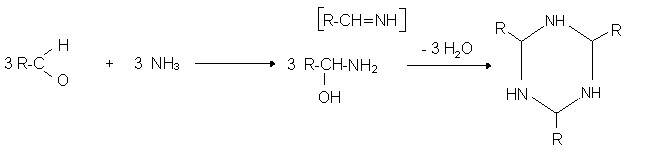
In the case of more complicated diamines, such as putrescines, we have two amine groups that also react with the aldehyde:
↑H2NCH2CH2CH2CH2NH2↑
Thus, the particularly unpleasant substances have a double “chance” to be neutralized. The equations presented are for illustrative purposes.za
If we assume that when the molecules of the two substances meet, a chemical reaction takes place as described above, and both compounds are at the minimum concentration to make them smellable, then one of the basic laws of chemistry says that the reaction will be “pushed” toward its completion by the substance that is in excess, that is, until there is (almost) no molecule of bad odour left.
Generally speaking, the reactions occurring take place at different rates depending on the type of compounds being neutralized. In rare cases, these reactions may be incomplete, requiring more time as well as some excess preparation. The environment pH has a severe impact on the reaction kinetics. If we consider the wastewater deodorization then the best results are achieved outside the pH extreme values.
Chemical reactions
Ammonia, amines and hydrogen sulphide are the most common odour air pollutants among the numerous groups of odours.
The active ingredients of WESTRAND’s preparations act by nucleophilic reactions with nitrogen and sulphur on the d+ carbon of the C=O group of aldehydes and ketones. In general, these reactions are neither fast nor complete in 100% until the neutralizing components appear in sufficient excess to push the reactions toward completion. The kinetics of these reactions is affected by the pH of the reaction environment and temperature.
Reactions of aldehyde with ammonia:
Reactions of aldehyde with amines:
- primary amines
- secondary amines
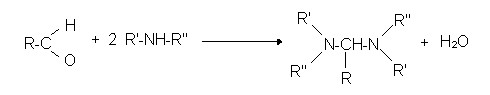
Reactions of aldehyde with mercaptans:
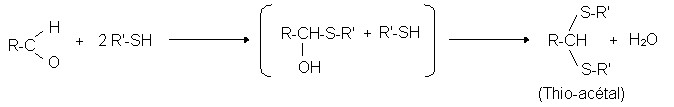
- H2S
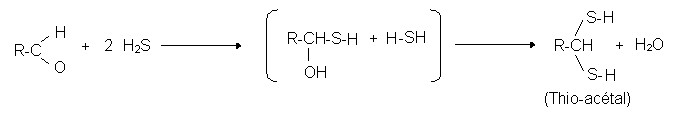
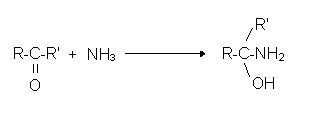
Reactions of ketones with ammonia:
Reactions of ketones with amines:
- primary amines
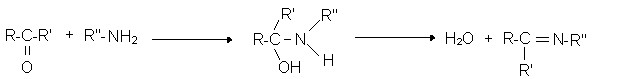
(Imines or Schiff’s principle)
- secondary amines
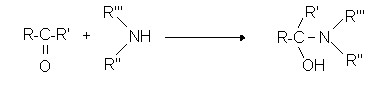
Reactions of ketones with mercaptans:
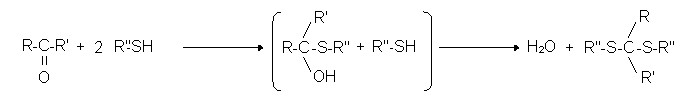
- H2S